TL;DR
Radioligand therapy in oncology is rapidly expanding, building on established targets such as PSMA in prostate cancer and SSTR2 in neuroendocrine tumors while exploring new areas like breast cancer and glioblastoma. The field is growing not because of new targets but due to advances in payloads, imaging, infrastructure, and operational know-how, making programs more reproducible and scalable. Historical approvals provide a blueprint, and next-generation alpha and beta emitters are accelerating differentiation. The current momentum reflects an industrialized, evidence-backed modality with high potential for clinical and commercial impact. The key for sponsors is pairing credible biology with operational readiness to scale beyond the modality’s original strongholds.
A modality with old approvals—and new momentum
Radioligand therapy is often described as “new,” but the concept is not. Radioimmunotherapies such as Zevalin (2002) and Bexxar (2003; later discontinued commercially) established early clinical precedent in lymphoma. More recently, peptide radioligands reshaped the field’s credibility with approvals in solid tumors: Lutathera (Lu-177 dotatate; 2018) for SSTR-positive GastroEnteroPancreatic NeuroEndocrine Tumors (GEP-NETs), and Pluvicto (Lu-177 vipivotide tetraxetan; 2022) for PSMA-positive metastatic castration-resistant prostate cancer (mCRPC), with an FDA label expansion in 2025.
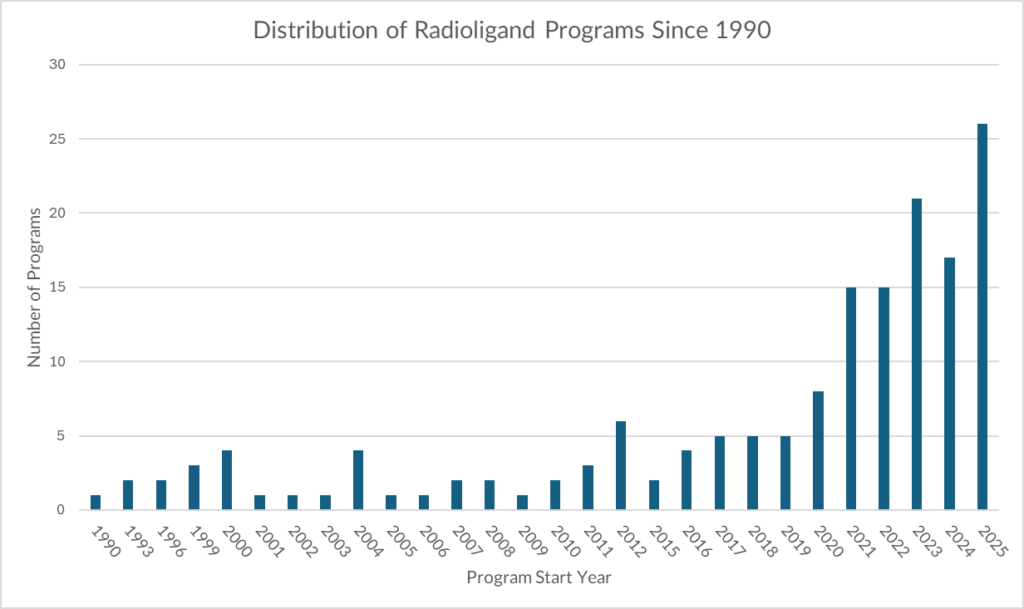
Against that backdrop, our dataset shows radioligand program initiation has accelerated sharply in the last few years, marking one of the clearest modality inflections currently visible in oncology development. Importantly, this growth appears to be driven less by a single “new approval moment” and more by a convergence of enablers: improved imaging, better payload chemistry, scaling infrastructure, and increasing sponsor confidence in a now-repeatable development pathway.
Why prostate and neuroendocrine tumors have dominated—and why that still matters
Historically, radioligand development has concentrated in two disease families: prostate cancer and neuroendocrine tumors (NETs). This has been consistent for a reason: both are unusually well-suited to theranostics.
- Target biology is unusually “clean.”
- PSMA expression in prostate cancer creates a high-contrast target for both imaging and therapy, enabling patient selection and response monitoring.
- SSTR2 expression in NETs similarly supports high-confidence targeting and has been clinically validated through Lutathera.
- PSMA expression in prostate cancer creates a high-contrast target for both imaging and therapy, enabling patient selection and response monitoring.
- Theranostics workflows are clinically actionable.
These diseases have benefited from a practical “scan → treat → rescan” model, making it easier to justify radioligands operationally and commercially relative to targets where expression is heterogeneous and imaging is less predictive. Radiotheranostics has become increasingly routine in prostate cancer and NET management, reinforcing the clinical logic of continued development concentration.
Emerging diversification: breast cancer and glioblastoma enter the frame
While prostate and NETs remain the foundation, our dataset shows diversification is now becoming visible, including increased attention to breast cancer and glioblastoma. This shift likely reflects two overlapping drivers:
- A search for next “radioligand-native” indications where tumor biology supports selective uptake (or where imaging can meaningfully de-risk selection).
- Competitive saturation in prostate pushing innovation toward adjacent tumor types while retaining familiar targeting principles (high expression, actionable imaging, defined patient subsets).
Breast cancer is also increasingly relevant to the radioligand story because it presents multiple accessible receptors and subtypes, and targets like gastrin-releasing peptide receptor (GRPR) have documented promise in prostate and breast contexts. Glioblastoma is a higher-risk extension, but it is consistent with sponsor behavior when infrastructure and platform confidence rise: high unmet-need tumors become testbeds for modalities seeking their next proof point.
Targets remain consistent—even as payloads evolve
A key pattern in the radioligand landscape is that targets remain comparatively stable, while payload strategies diversify. In our dataset:
- PSMA remains dominant, reflecting prostate leadership and strong precedent.
- SSTR2 is the next major anchor via NETs and validated clinical pathways.
- GRPR follows as a well-established theranostics and imaging target—especially relevant in prostate, with growing attention across other cancers.
This is strategically important: it suggests the modality’s growth is not being driven by constant reinvention of biology, but by maturing execution around a handful of credible targets—an adoption pattern typically associated with scalable platforms.
Payloads are where the innovation is accelerating
While targets in radioligands remain relatively stable (PSMA, SSTR2, GRPR), the payload layer—the radioisotope and its radiochemistry—has become the main axis of innovation. Historically, the clinical and commercial backbone of radioligand therapy was built on beta emitters, particularly Lu-177 (e.g., Pluvicto; Lutathera) and earlier Y-90 radioimmunotherapy (e.g., Zevalin), where the balance between penetration depth, dosimetry, and tolerability is better understood. The current wave is increasingly exploring next-generation payload strategies, especially alpha emitters (e.g., Ac-225, Pb-212, Th-227), aiming to improve potency and tumor cell kill in settings where beta-emitter efficacy may plateau. In our dataset, beta-emitter payloads still represent the majority of active programs, but alpha-emitter programs are the fastest-growing subset.
This payload evolution matters for two reasons. First, it explains how sponsors can differentiate within the same target: two PSMA programs may look similar on biology, but diverge substantially on therapeutic index, safety profile, and positioning depending on isotope choice and linker/chelator design. Second, it reflects a broader maturation of the field’s “engineering layer”: improved chelation chemistry, linker stability, and dosimetry planning are making radioligands more reproducible and scalable across sites. In other words, much of the recent radioligand momentum is not because targets are newly discovered—it is because payload and delivery science has expanded what is feasible.
The approval caveat: few assets, meaningful precedent
The historical approvals underpinning this modality remain limited in count. In the past, eight approved programs across four assets—Bexxar, Zevalin, Pluvicto, and Lutathera—established the approval precedent in lymphoma subtypes, NETs (including pancreatic/GEP-NETs), and prostate cancer.
The implication is two-sided: radioligands have fewer approvals than mainstream modalities, but they have enough validated examples in high-visibility diseases to create a credible blueprint—particularly now that modern radiotheranostic workflows are more standardized.
Why growth is accelerating now: what has changed
The recent wave of radioligand programs is best understood as the result of multiple constraints being removed at once. Earlier radiopharmaceutical efforts proved the science could work, but they struggled to scale in a way that fit modern oncology drug development. The current acceleration reflects a field that has become more “industrialized”: more predictable manufacturing, clearer clinical workflows, and more standardized evidence generation.
1) Infrastructure has moved from niche to scalable
Radioligand therapy used to be limited by the availability of appropriately licensed sites, trained staff, radiopharmacy capacity, and radiation safety workflows. That bottleneck is easing. The number of centers capable of administering therapeutic radiopharmaceuticals has expanded, and many health systems have become more comfortable operationalizing “theranostic” pathways that combine imaging-based identification with treatment delivery. This matters because radioligands are not merely drugs—they require an ecosystem (radiopharmacy, nuclear medicine, scheduling, waste management, compliance) that must function reliably at volume. Recent analyses emphasize the growth of theranostics capacity and procedure volume, including Lu-177 PSMA therapy.
2) Manufacturing and supply chains are more credible than they were
Historically, supply risk was a real deterrent: isotope production, short half-lives, and cold-chain constraints created uncertainty for both trials and commercial planning. Over time, the supply base has broadened and become more integrated. Sponsors now plan radiopharmaceutical programs with a clearer view of isotope sourcing, centralized vs. distributed manufacturing, and regional distribution strategies. As supply risk becomes more manageable, investment committees become more willing to greenlight programs that would previously have been viewed as “operationally fragile.”
3) The field has a clearer clinical blueprint than a decade ago
Radioligands are no longer a purely exploratory modality. Prostate and neuroendocrine tumors have established a repeatable development template: select patients through target-positive imaging, dose and schedule around known radiobiology, and generate outcomes that map to accepted endpoints. That blueprint is reinforced by high-visibility approvals in modern theranostics such as Lutathera and Pluvicto.
Crucially, a blueprint reduces execution risk: trial designers are not inventing workflows from scratch, sites are familiar with endpoints and logistics, and regulators have precedent for review.
4) Imaging has matured into a de-risking tool, not just a diagnostic adjunct
A major reason radioligands “fit” prostate and NETs is the ability to confirm target expression noninvasively and dynamically. The field has increasingly adopted imaging as a formal gating mechanism—filtering for target-positive disease, monitoring response, and supporting clinical narratives. This can reduce failure rates by ensuring that trials enroll patients biologically positioned to benefit. Reviews of radiotheranostics emphasize how imaging integration improves patient selection and development confidence.
5) Payload and chemistry advances have widened the design space
Modern radioligand programs benefit from improved linker chemistry, chelators, and formulation approaches that can improve stability and therapeutic index. The payload landscape is also broadening: while beta emitters remain foundational, there is rising interest in next-generation approaches (including alpha emitters) where the risk-reward calculus may be compelling for resistant disease or micrometastatic settings. This growing toolkit enables sponsors to iterate while keeping targets constant—one reason the target landscape remains stable even as program volume rises.
6) Earlier commercial failures taught the industry what not to do
The case of Bexxar is often cited as an example where scientific value did not translate into sustained adoption, due to referral patterns, workflow complexity, and reimbursement logistics. The modern wave is building with those lessons in mind: programs increasingly incorporate delivery planning early, align with imaging infrastructure, and pursue indications where theranostic workflows are already accepted. In other words, the modality’s resurgence is not a repeat of the early 2000s—it is a second cycle, shaped by operational learning and clearer market integration.
Bottom line: radioligands are accelerating now because the modality has crossed a threshold from “can it work?” to “can it be delivered, repeated, and scaled?” That shift—more than any single approval—best explains the current program explosion.
| Category | Overall Historical Approval Rate | Number of Historic Programs |
| Overall Oncology | 6% | 13023 |
| Radioligands | 13% | 62 |
| Monoclonal Antibodies (mAbs) | 6% | 4857 |
| Small Molecule Inhibitors (SMIs) | 6% | 8401 |
| Cell therapies | 6% | 410 |
| Antibody-drug Conjugates (ADCs) | 6% | 697 |
The dataset indicates superior historical approval performance for radioligands relative to several well-known modalities, but the denominator is small. That combination—high apparent success with limited precedent—supports enthusiasm, but also raises the importance of execution discipline as the modality expands beyond its “native” strongholds.
What to watch next
The radioligand surge appears structurally supported, not purely sentiment-driven. However, the next phase of the modality will be decided by whether expansion beyond prostate/NETs can reproduce the same theranostic clarity: target expression robustness, imaging predictiveness, and scalable delivery.
Breast cancer and glioblastoma represent two distinct tests: one a logical receptor-rich expansion arena, the other a high-risk unmet-need frontier. The winners will likely be sponsors that pair credible biology with operational readiness—because in radioligands, the platform is only as strong as the system that delivers it.
How we support decision-makers in radioligand strategy
Clinical development teams: We contextualize radioligand programs against historical modality performance, highlight where target/payload choices align with proven pathways, and benchmark how similar programs progressed by phase and indication.
Portfolio strategists: We quantify modality momentum versus maturity—helping leaders separate scalable trends from crowded bursts, and prioritize where evidence and infrastructure are converging.
BD and licensing leads: We provide fast competitive context—who is building on validated targets versus who is stretching into higher-risk territory—and frame differentiation in terms that matter for late-stage feasibility and adoption.
Radioligands are moving from niche to platform. The question now is not whether the modality can work—it is where it can scale next.
For more tailored, data-rich insights, let’s talk.


