Intravenous, Oral, and Subcutaneous Development Patterns Across Oncology, I&I, Neurology, and Metabolic Disease
TL;DR
Route of administration has evolved into a strategic lever that influences development, adoption, and lifecycle value. Its role varies by therapy area: oncology remains IV-led with subcutaneous mainly as a lifecycle tool, immunology has shifted toward subcutaneous biologics, neurology is structurally oral, and metabolic diseases are increasingly driven by subcutaneous peptides. Higher approval rates for subcutaneous programs often reflect later-stage, de-risked use rather than inherent advantage. Ultimately, the right route is the one that best fits biology, care delivery, and commercial strategy.
Why routes of administration are no longer a “formulation detail”
Route of administration (RoA) has moved from an operational afterthought to a strategic lever. Across therapeutic areas, it influences trial feasibility, site capacity, patient experience, adherence, payer management, and—critically—product lifecycle defensibility. RoA shifts are also increasingly used as competitive tactics, particularly when a molecule’s core mechanism is no longer unique.
Our clinical-program benchmarks show that the “RoA story” is not uniform across therapy areas. Each has its own physics—driven by disease severity, treatment setting, safety tolerance, delivery infrastructure, and the degree to which patient convenience can be monetized.
| Category | Historical Approval Rate |
| Overall Oncology | 6% |
| Oncology Intravenous | 6% |
| Oncology Oral | 6% |
| Oncology Subcutaneous | 12% |
| Overall Immunology | 14% |
| Immunology Intravenous | 13% |
| Immunology Oral | 8% |
| Immunology Subcutaneous | 23% |
| Overall Neurology | 16% |
| Neurology Intravenous | 19% |
| Neurology Oral | 16% |
| Neurology Subcutaneous | 16% |
| Overall Metabolic | 14% |
| Metabolic Intravenous | 9% |
| Metabolic Oral | 11% |
| Metabolic Subcutaneous | 22% |
Oncology: IV still dominates—SC grows, but remains structurally constrained
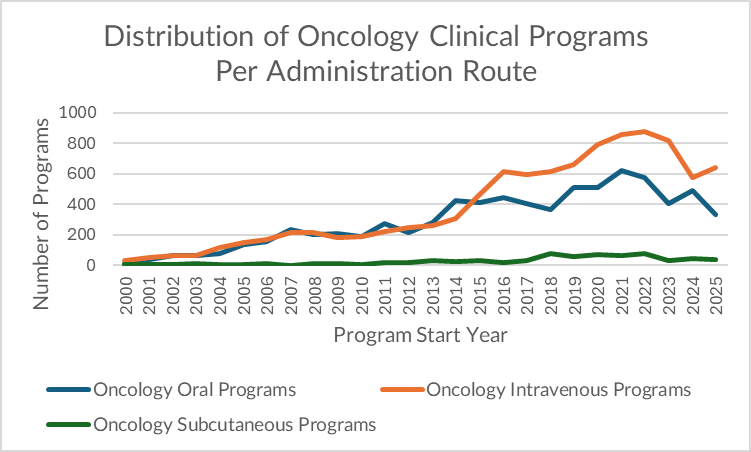
Oncology remains an IV-first ecosystem. In 2025 alone, our dataset shows 639 IV oncology programs versus 335 oral and just 36 subcutaneous (SC) programs. IV remains the default because oncology trials prioritize:
- Dose control and rapid titration, particularly for therapies with narrow therapeutic windows or complex PK/PD.
- Complex regimens and combinations (e.g., IO + chemo, multi-agent protocols) that fit infusion center workflows.
- High acuity patient management, including infusion reactions and intensive monitoring, which are operationally easier in supervised IV settings.
Despite years of industry chatter about SC, the volume reality is clear: SC is still the smallest RoA channel in oncology.
Yet the SC narrative persists for a reason. The commercial rationale is powerful: SC can reduce chair time and staff burden materially, which matters in increasingly capacity-constrained infusion environments. Published analyses show large reductions in chair time and healthcare professional time when moving from IV to SC for certain biologics.
The second reason: SC is increasingly a lifecycle management and market-defense tool, not merely a patient-convenience upgrade. A high-profile example is Keytruda’s new SC formulation, approved by FDA in 2025, explicitly positioned around speed of administration (minutes rather than a 30-minute infusion) and adoption expectations.
Why oncology SC is “real,” but not a primary development route
A key pattern in oncology is that many SC approvals are not “new drugs”—they are new formulations of previously successful IV agents. That sequencing is logical: converting to SC often comes after the molecule has already proven efficacy and safety, and after the sponsor can justify large-scale formulation, device, and bioequivalence investment. This is why SC looks strategically important even while it remains small in raw program counts.
What the HAR table reinforces:
- Overall Oncology HAR: 6%
- Oncology IV HAR: 6%
- Oncology Oral HAR: 6%
- Oncology SC HAR: 12%
At face value, SC appears to outperform. But the interpretation is nuanced: because SC oncology programs are frequently later-lifecycle, de-risked conversions of proven assets, the approval rate advantage is directionally plausible—yet not evidence that “SC works better” as an early development choice. It is best read as: once a drug works, converting it to SC can be an additional path to value.
Assessment: In oncology, SC is not a fad—but it is also not a wholesale modality shift. It is a selective, commercially motivated trend driven by capacity constraints, patient experience, and patent/competition strategy—especially for high-volume biologics.
Immunology & Inflammation: the platform shift is already underway
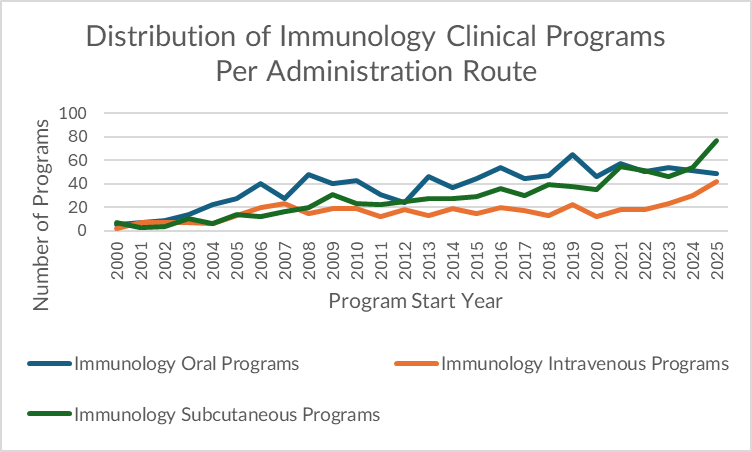
I&I has undergone the opposite evolution. Historically, oral therapies dominated early, because many inflammatory diseases were managed long-term and systemically with small molecules—where daily dosing and chronic use fit oral delivery. But today, SC is the leading route for new program initiations, and IV is increasingly competitive behind it.
In 2025, our dataset shows I&I programs split as: 77 SC vs 49 oral vs 42 IV. This distribution reflects the reality that modern I&I has become a biologics-first market in several major indications.
Why SC now leads in I&I:
- Biologics have become standard in multiple high-prevalence diseases (e.g., RA, psoriasis, IBD).
- SC enables home administration or minimal-touch settings—valuable for chronic diseases and resource-constrained systems.
- The field has matured around delivery technologies (including hyaluronidase-enabled SC administration), making SC broadly feasible at scale.
HAR strengthens the thesis:
- Overall I&I HAR: 14%
- I&I Oral HAR: 8%
- I&I IV HAR: 13%
- I&I SC HAR: 23%
Here, SC’s advantage is less likely to be “conversion bias” alone; I&I biologics are frequently designed for SC from early stages, so SC performance may reflect both regulatory maturity and execution feasibility in chronic diseases.
Neurology: oral dominance is structural, not cyclical
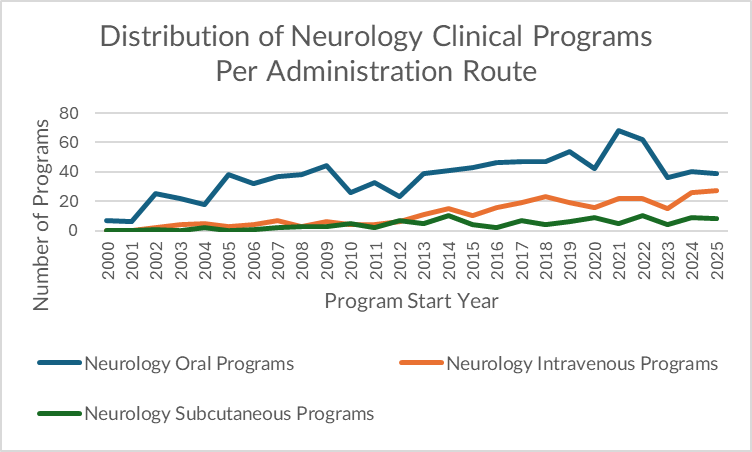
Neurology stands out for consistency. Oral has dominated across the entire historical curve, and IV/SC trail steadily behind. In 2025: 39 oral vs 27 IV vs 8 SC.
Why oral dominates neurology:
- Many neurological therapies are chronic and adherence-dependent, making outpatient oral dosing the default.
- Several neurology indications require long-term disease control rather than episodic intervention—favoring oral formats.
- For CNS-active drugs, development often starts with small molecules designed for systemic exposure and CNS penetration. (For MS specifically, oral therapies have been positioned as major disease-modifying options for years.)
The HAR table suggests neurology is the most route-agnostic in outcomes:
- Overall Neurology HAR: 16%
- Neurology IV HAR: 19%
- Neurology Oral HAR: 16%
- Neurology SC HAR: 16%
This is consistent with a TA where route is often determined by drug class and target biology (e.g., antibodies and infusions in certain diseases), but oral remains the broad default.
Metabolic: SC has overtaken oral—driven by the modern peptide era
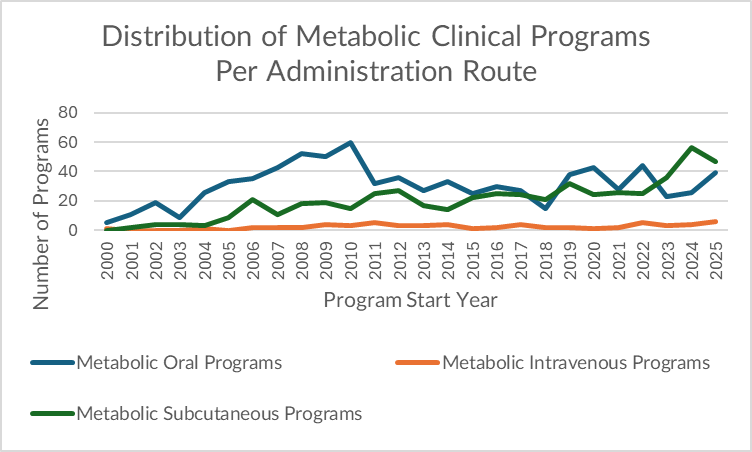
Metabolic has fewer programs overall, but the RoA shift is clear. Oral used to dominate (reflecting legacy T2D and cardiometabolic small molecules), but SC now leads, and IV remains low and stable. In 2025: 47 SC vs 39 oral vs 6 IV. SC peaked in 2024 (56) and remains elevated.
The reasons are less mysterious than they appear: the metabolic market has been reshaped by injectable peptide therapies (GLP-1 and beyond), where SC has become the standard delivery route for efficacy, tolerability, and dosing control.
HAR supports the shift:
- Overall Metabolic HAR: 14%
- Metabolic Oral HAR: 11%
- Metabolic IV HAR: 9%
- Metabolic SC HAR: 22%
As in I&I, SC’s higher approval performance is directionally consistent with a TA where the most successful modern mechanisms are delivered subcutaneously.
The cross-TA pattern: SC is winning where biology is biologic- or peptide-led
Across the four therapy areas, SC grows fastest where development is increasingly driven by biologics/peptides and long-term chronic use. Where the therapeutic model is acute, combination-heavy, and site-supervised—as in oncology—IV remains dominant, and SC appears primarily as a post-proof lifecycle lever.
A practical way to summarize the evidence:
- Oncology: IV-first; SC as lifecycle optimization, not primary strategy.
- I&I: SC is already the default for new biologics; oral is losing share.
- Neurology: oral dominance is stable; IV/SC remain secondary.
- Metabolic: SC has become the growth engine of modern programs.
How we support decision-makers designing for the “route reality”
Our benchmarking and analytics work helps teams evaluate RoA decisions as part of program strategy—not packaging.
- For clinical development leaders, we contextualize route choices against historical transition and approval patterns by TA, helping teams avoid route-driven feasibility traps and align endpoints and settings with how care is actually delivered.
- For portfolio strategists, we quantify where SC momentum reflects genuine platform adoption (I&I, metabolic) versus selective lifecycle maneuvering (oncology), supporting smarter prioritization and investment timing.
- For BD and licensing teams, we assess whether route claims represent real differentiation or a late-stage conversion play—critical for valuation in crowded classes where convenience can be monetized.
The route of administration is now a competitive strategy. The winners will be those who pick routes that fit the biology, the care pathway, and the commercial lifecycle—not just the narrative.
For more tailored, data-rich insights, let’s talk.



